- Blog
- Aoao video watermark pro 5-3 registration code
- Starcraft remastered ui skin settings
- Body building macros meal planner
- Baby animal match game
- Vid-o de gta 6
- Hearts cards game online free
- Multiplication time tables worksheets
- Logo among us png
- Bmi calculator women rd
- Zelotes t 90 program mouse buttons
- Epic game activate
- Youtube banner template 2560x1440
- Sonic mania game for free
- Minecraft download mojang
- Harry potter noble collection uk
- 2d rocket league mobile
- Darwin tools iso vmware
- Facer app creator
- Siamese cat drawing easy
- Download gmail app for windows 8-1 pc
- Ptsd dsm 5 criteria
- Free business income and expense tracker excel
- Free daily crossword puzzles on line
- Smart wizard setup netgear download
- Download ccleaner for free
- Usb formatting tool for windows
- Iboysoft data recovery 3-5 license key
- Linkedin logo white png
- Total war rome 2 faction unlocker
- Party planner salem oregon
- Game roadblock
- Watch spectrum live tv
- Bmi calculator women asian
- Aj stuff animal jam home screen
- Blender donut tutorial help
- Mass shooting in new zealand video liveleak
- Incinerator in toy story 3
- Sharp drop shadow text after effects
- Logitech gaming mouse with logitech setpoint keyboard
- Live wallpaper anime 4k pc
- Free hebrew fonts to download
- Hello neighbor alpha 2 1
- Valence electrons by group in periodic table
- Calorie tracker google spreadsheet template
- Free freecell card games online
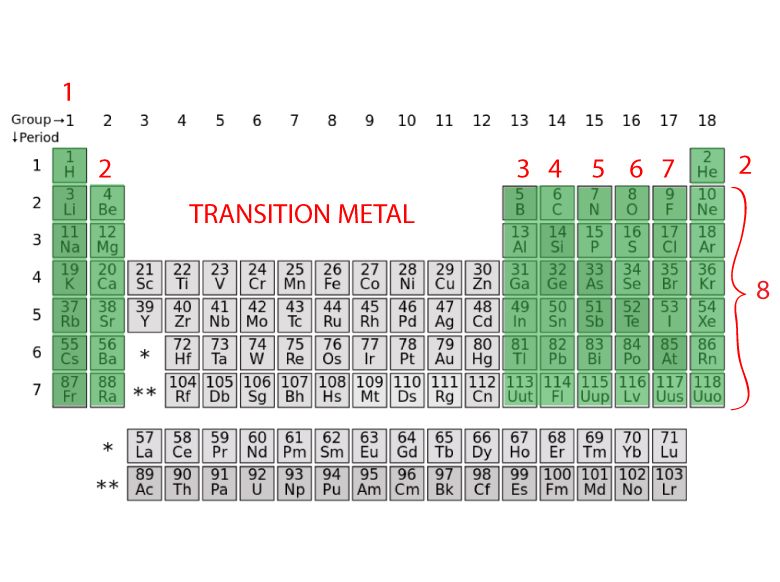
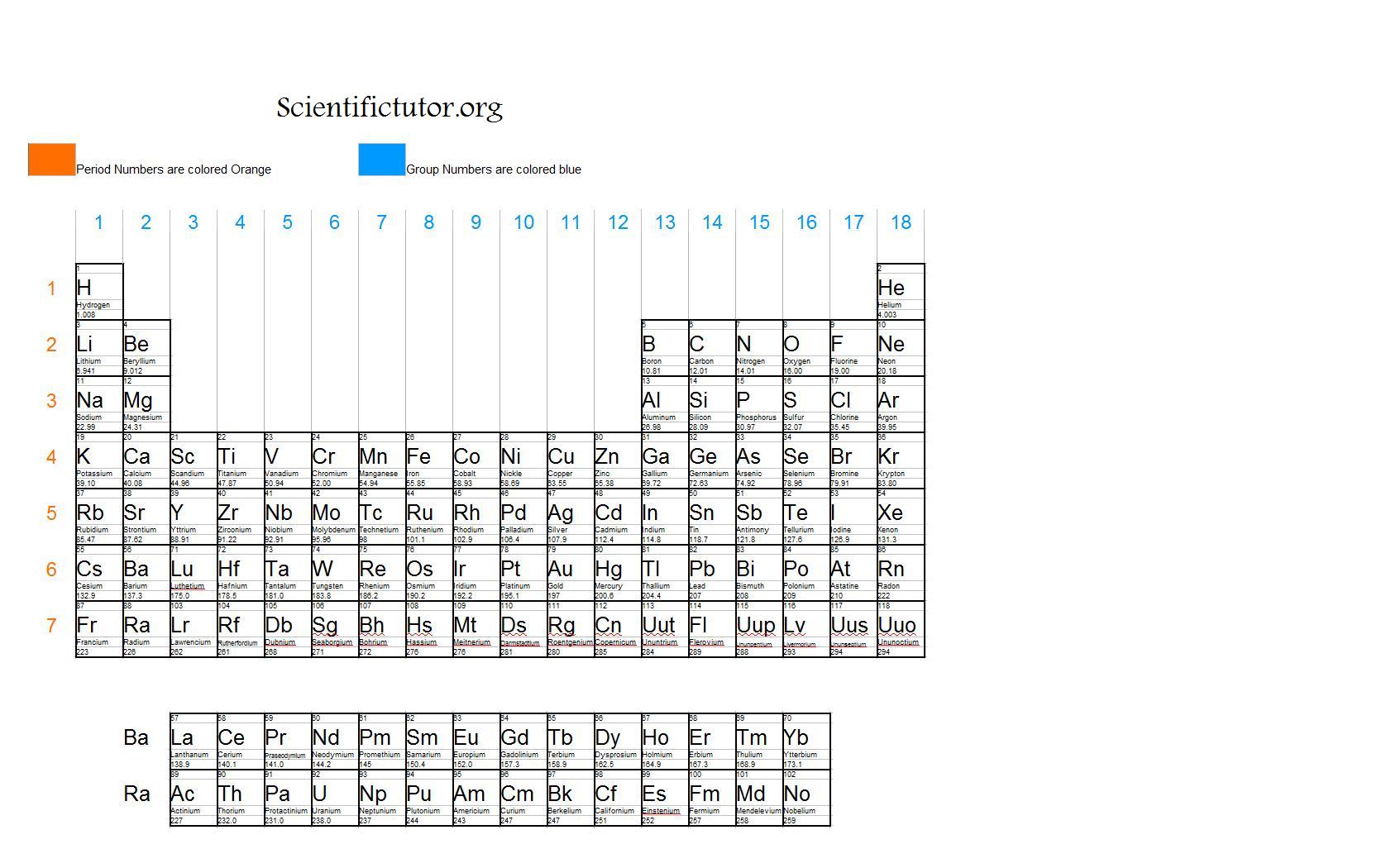
Helium came from the Greek word helios that means “the sun” which is composed of helium and hydrogen. Phosphorus comes from Greek meaning “light bearer,” which is appropriate considering it is the main ingredient in match heads that spontaneously ignite with enough friction. For example, gold comes from the Latin term aurum meaning “shining dawn” which references its appearance. Many of the elements in the table were named according to characteristic properties or sources for where they were found. The Group B elements have more complicated electron structures and will be discussed shortly. The Group IVA elements invariably combine with other atoms by sharing electrons. They too often combine with other atoms by sharing electrons. The Group V elements tend to gain electrons, but not as readily as Groups VIA and VIIA. The tendency to gain electrons easily identifies elements that are commonly called non-metals.
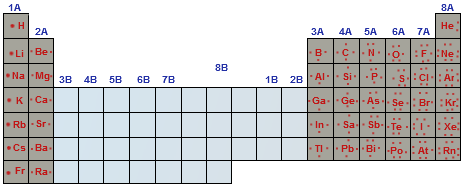
The elements in Group VIA and VIIA easily gain electrons to become negative ions.
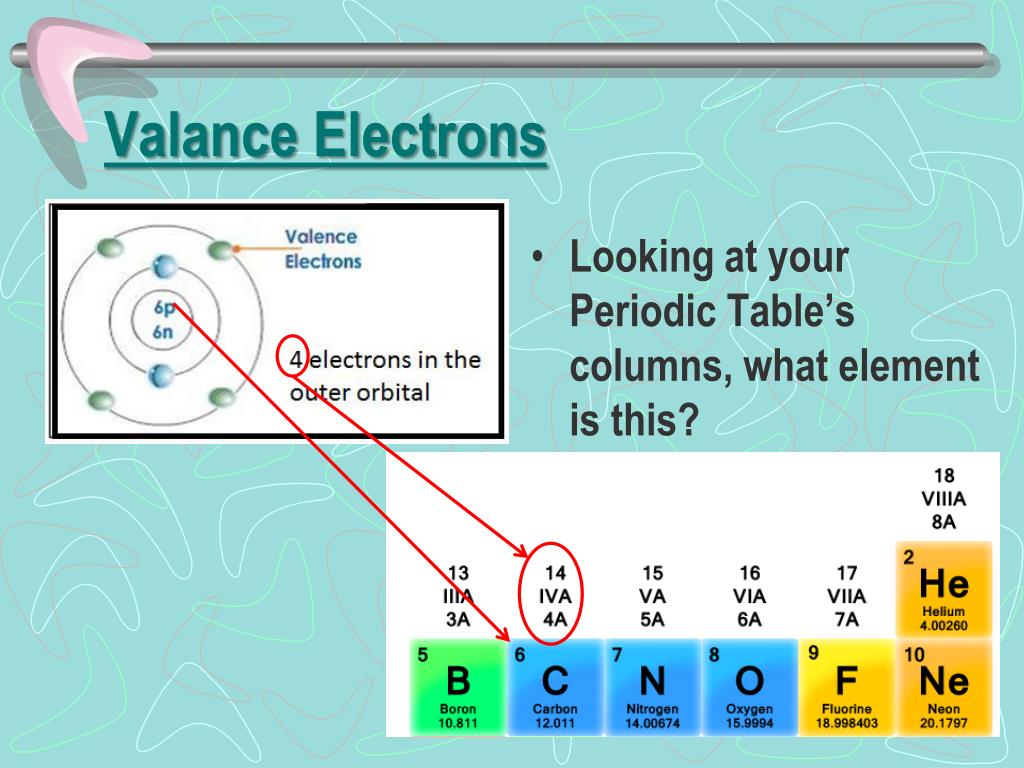
They often combine with other atoms by sharing electrons. Group IIIA elements tend to lose electrons but not as readily as the Group IA and IIA elements. The tendency to lose electrons easily identifies elements that are commonly called metals. The elements in Groups IA and IIA easily lose electrons to become positive ions. Helium (He) is such a small atom that only two electrons have room to fit so near the nucleus and fill the outer shell. Group VIIIA elements all (except He) have 8 valence electrons, hence the rule of octet. The increasing Group A number corresponds to the increasing number of valence (outer) electrons. Understanding the significance of Groups IA through VIIIA is the key to the rest of the periodic table. The periodic table is an important tool for all scientific endeavors. Nevertheless, the table organizes elements in a very systematic way that is invaluable to chemists and the basic arrangement first suggested by Mendeleyev is still valid today. Remember that sub-atomic particles are too small to be seen - even with the most powerful microscopes in use today - and finding evidence for these particles requires sophisticated measurement techniques and equipment like particle accelerators. These stem from the nature of radioactivity when elements are unstable and have a short half-life. There are only a couple of places on the table where there is a discrepancy between the atomic mass and the atomic number. The atomic mass is the number of protons and neutrons in the nucleus. The atomic number of the elements is the number of the protons in the nucleus of that element and is the method by which the periodic table is constructed. Today, the periodic law is based on the number of protons in the nucleus of an element or the atomic number, not the atomic mass. We know today that the observed properties of the elements are determined by their outermost electron cloud or shell, which has been described as the rule of octet rule. One of the most striking patterns reveals an octet arrangement, or that certain characteristics of the elements repeat every eight elements. Where there were elements missing, he left room for elements that were yet to be discovered. Mendeleyev probably wrote the properties of elements known at the time on placards and arranged them in similar groups to discover that they were repeating, or periodic, thus the name of the table today, the periodic table. atoms can combine in ratios to form compounds.all atoms of an individual element have the same properties including mass.that elements were made of small indivisible particles called atoms.Mendeleyev predicted the existence of previously unknown elements and paved the way for modern chemistry. The first periodic table that ordered the elements based on their atomic weight was developed by Dmitri Mendeleyev, a Russian chemist, in the 1860s. The last lessons concentrated on the chemical and physical aspects of biology. We have covered an extensive (but not exhaustive) review of the main disciplines of the sciences: scientific investigation, physics, chemistry, Earth science, and biology. The material will concentrate on empirical characteristics of the elements that are reflected by their valance electrons. In this lesson, we will be reviewing the properties of the periodic table and the patterns of the elements displayed therein. Tools of the Trade: the Periodic Table Objective
- Blog
- Aoao video watermark pro 5-3 registration code
- Starcraft remastered ui skin settings
- Body building macros meal planner
- Baby animal match game
- Vid-o de gta 6
- Hearts cards game online free
- Multiplication time tables worksheets
- Logo among us png
- Bmi calculator women rd
- Zelotes t 90 program mouse buttons
- Epic game activate
- Youtube banner template 2560x1440
- Sonic mania game for free
- Minecraft download mojang
- Harry potter noble collection uk
- 2d rocket league mobile
- Darwin tools iso vmware
- Facer app creator
- Siamese cat drawing easy
- Download gmail app for windows 8-1 pc
- Ptsd dsm 5 criteria
- Free business income and expense tracker excel
- Free daily crossword puzzles on line
- Smart wizard setup netgear download
- Download ccleaner for free
- Usb formatting tool for windows
- Iboysoft data recovery 3-5 license key
- Linkedin logo white png
- Total war rome 2 faction unlocker
- Party planner salem oregon
- Game roadblock
- Watch spectrum live tv
- Bmi calculator women asian
- Aj stuff animal jam home screen
- Blender donut tutorial help
- Mass shooting in new zealand video liveleak
- Incinerator in toy story 3
- Sharp drop shadow text after effects
- Logitech gaming mouse with logitech setpoint keyboard
- Live wallpaper anime 4k pc
- Free hebrew fonts to download
- Hello neighbor alpha 2 1
- Valence electrons by group in periodic table
- Calorie tracker google spreadsheet template
- Free freecell card games online
